Sunscreen Innovation Act Signed Into Law
12 December, 2014
Between the Thanksgiving holiday and Black Friday shopping, an important bit of Executive Branch news flew under the radar last week. After being passed earlier in the year by both the House and the Senate, President Obama signed the Sunscreen Innovation Act into law on Nov. 29. The law is intended to clear the more than decade-long backlog of sunscreen ingredients awaiting FDA approval. Some ingredients awaiting this approval are more effective at blocking UVA rays from the sun, and most have been in use in Europe for years.
In a July article in Time, ACS CAN President Chris Hansen said, “Educating people about good sun safety behaviors is only half of the story. American consumers should have access to the broadest choice of sunscreens — including those in use for years in other countries — once they are shown to be safe and effective.”
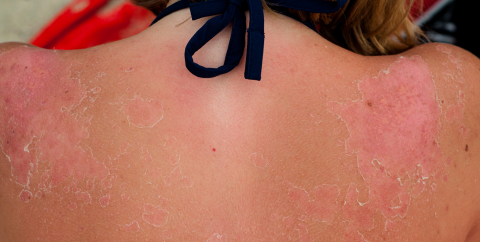
According to the U.S. Surgeon General’s Call to Action to Prevent Skin Cancer, skin cancer is the most commonly diagnosed cancer in the United States, and most cases are preventable. Most skin cancers are at least partially caused by UV exposure, so reducing exposure reduces skin cancer risk. In Nevada, many counties are above the national average in incidence for skin cancer and melanoma.
Read more about skin cancer and melanoma here.
Get more information on the U.S. Surgeon General’s Call to Action to Prevent Skin Cancer.
Read Time’s article from this week on the President’s signing.
photo credit: SVTHERLAND via photopin cc
You May Also Like

Federal Funding Supports Cancer Control in Nevada
02.04.2025
The change of presidential administrations in January 2025 brought with it increased scrutiny of federal program funding.

Fall into Sun Safety: Why Wide-Brimmed Hats and Sunscreen Are Autumn Essentials
11.04.2024
As the leaves begin to change and temperatures cool, it's easy to forget about sun protection.
Skin cancer continues to be most common cancer in Nevada, U.S.
05.01.2023
More people are diagnosed with skin cancer each year in Nevada than all other cancers combined, according to the American Can